Bladder Cancer Report
: Analysis on the Market, Trends, and TechnologiesThe bladder cancer landscape is shifting toward diagnostic-led pathway control, anchored by a 2024 market size of $266,800,000 and a CAGR of 4.4%, with forecasts reaching $345,300,000 by 2030
This report was last updated 106 days ago. Spot an error or missing detail? Help us fix it by getting in touch!
Topic Dominance Index of Bladder Cancer
To identify the Dominance Index of Bladder Cancer in the Trend and Technology ecosystem, we look at 3 different time series: the timeline of published articles, founded companies, and global search.
Key Activities and Applications
- Non-invasive early detection and surveillance: Development and commercial deployment of urine-based methylation and multi-omic assays to replace or triage routine cystoscopy in low-risk cohorts, reducing procedure volume and cost per patient.
- Risk-stratified surveillance: Using molecular classifiers and ctDNA to segment NMIBC patients into differential surveillance frequencies and intervention thresholds, lowering lifetime surveillance cost and improving patient experience.
- Neoadjuvant and bladder-sparing immuno-combination regimens: Clinical programs pairing checkpoint inhibitors or ADCs with localized agents to increase pathological complete response and expand organ-preserving options for muscle-invasive disease Merck Advances Treatment of Bladder and Kidney Cancers.
- Sustained intravesical drug delivery: Commercialization of hydrogel and depot systems that maintain therapeutic concentrations in the bladder wall for weeks, enabling outpatient, non-surgical tumor control for select NMIBC populations.
- AI-assisted endoscopy and optical biopsy: Integration of machine-learning lesion detection with blue-light and OCT/optical biopsy platforms to reduce sampling error and guide resection margins in real time Photocure ASCO GU reporting; Scinvivo technology overview.
Emergent Trends and Core Insights
- Epidemiology vs. economics: Global incidence remains high (≈614,300 new cases; 220,600 deaths in 2022) while age-standardized rates slowly decline in some markets—creating growing absolute case volumes that sustain demand for both diagnostics and repeated interventions Global trends in the epidemiology of bladder cancer.
- Diagnostics as the commercial gatekeeper: High-sensitivity urine assays and methylation panels are moving from adjunct to primary triage tools; regulatory and reimbursement moves (e.g. national reimbursement decisions and Breakthrough Device designations) materially accelerate adoption in surveillance pathways.
- Therapy bifurcation: Investment concentrates on either (a) augmenting the BCG backbone with immune activators and oncolytics for BCG-unresponsive disease, or (b) deploying localized pharmacokinetic solutions (gels, nanoparticles, ultrasound-driven delivery) to achieve high intravesical exposure with lower systemic toxicity UGN-102/RTGel programs in pipeline analyses.
- Reimbursement and guideline gating: Payer coverage decisions for urine assays and outpatient intravesical devices will determine which diagnostic and delivery players scale; early national reimbursement decisions (e.g. Netherlands for select methylation tests) create regional beachheads.
- Operational shift to outpatient models: Blue-light cystoscopy, longer-dwell intravesical formulations, and ASC adoption compress hospital utilization and shift revenue mix toward device consumables and recurring diagnostics Mordor Intelligence market dynamics.
Technologies and Methodologies
- Methylation-based liquid biopsy panels: PCR and NGS workflows for urine methylation signatures that enable recurrence detection and prognostic stratificationPangea Laboratory.
- RTGel sustained-release intravesical platforms: Reverse-thermal hydrogels that extend local drug exposure (enabling outpatient instillation with multi-week dwell) and support non-surgical tumor clearance programs.
- Optical biopsy and OCT imaging: Forward-looking OCT catheters and enhanced visualization tools that reveal subsurface invasion depth and tumor margins intra-procedurally, reducing under-resection rates.
- Oncolytic and microbial immunotherapies: Intravesical oncolytic vectors and BCG-based delivery of immune agonists (e.g. STING, IL-15 agonists) designed to convert local immunogenicity into durable responses for BCG-resistant NMIBC CG Oncology.
- AI-enabled image analytics and decision support: Real-time lesion detection during cystoscopy and post-procedure pathology triage algorithms that reduce operator variability and optimize biopsy targeting.
- Nanoparticle and liposomal local delivery: Liposomal formulations and neoliposomal carriers for intravesical chemotherapeutics (e.g. targeted paclitaxel preparations) to amplify intratumoral exposure and lower systemic toxicity LIPAC Oncology.
Bladder Cancer Funding
A total of 226 Bladder Cancer companies have received funding.
Overall, Bladder Cancer companies have raised $26.9B.
Companies within the Bladder Cancer domain have secured capital from 975 funding rounds.
The chart shows the funding trendline of Bladder Cancer companies over the last 5 years
Bladder Cancer Companies
- Scinvivo B.V. — Scinvivo develops an optical biopsy platform (forward-looking OCT) that images beneath the bladder surface to determine invasion depth and tumor borders during cystoscopy; this reduces unnecessary resections and improves margin control. The technology targets procedure accuracy gains that directly lower recurrence and repeat-procedure costs. The company is progressing CE/FDA pathing and emphasizes integration with existing cystoscope workflows.
- OncoSTING — OncoSTING is advancing a BCG-based delivery of STING agonists to potentiate local immune activation for NMIBC, pursuing an approach that directly tackles BCG-unresponsive patients. Their R&D focuses on combining established intravesical vectors with novel immunostimulants to raise durable response rates while maintaining acceptable safety profiles.
- Trigone Pharma — Trigone Pharma engineers sustained-release local delivery systems for bladder diseases with a sustained-release product pipeline aimed at BCG-refractory NMIBC and other urologic targets. Their platform seeks to extend dwell time and reduce administration frequency, enabling outpatient management and improved patient adherence.
- Cystotech — Cystotech provides AI-driven decision support tools for cystoscopic imaging and treatment planning, focusing on automated lesion detection and risk scoring to standardize reporting and reduce clinician variability in surveillance pathways. The software is designed to complement visualization platforms rather than replace them, fitting into existing clinical workflows.
- Bladderly — Bladderly offers an AI-powered digital bladder diary that passively captures urinary volumes and symptoms via sound analytics, enabling longitudinal symptom tracking and remote triage for hematuria referral pathways. The solution aims to improve patient adherence to surveillance and collect real-world outcome signals that can enrich registries and payer evidence packages.
Identify and analyze 1.2K innovators and key players in Bladder Cancer more easily with this feature.
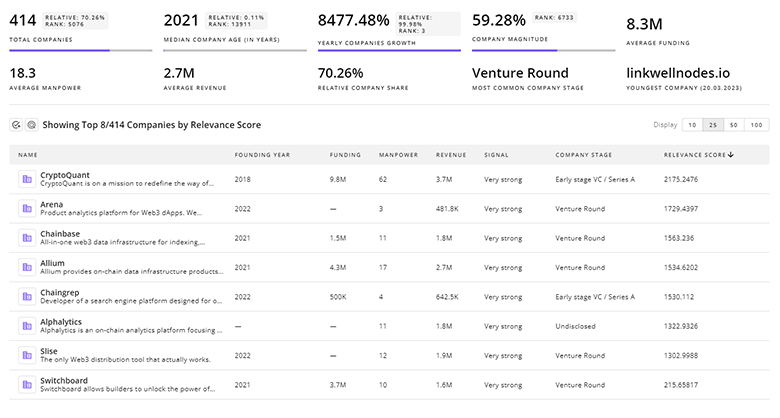
1.2K Bladder Cancer Companies
Discover Bladder Cancer Companies, their Funding, Manpower, Revenues, Stages, and much more
Bladder Cancer Investors
TrendFeedr’s investors tool offers a detailed view of investment activities that align with specific trends and technologies. This tool features comprehensive data on 1.0K Bladder Cancer investors, funding rounds, and investment trends, providing an overview of market dynamics.
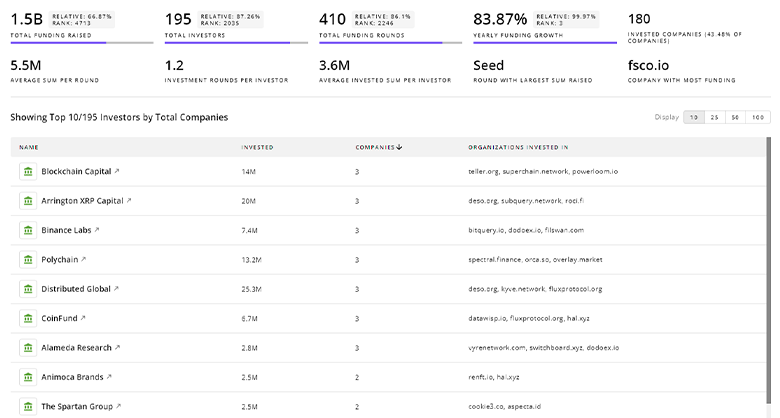
1.0K Bladder Cancer Investors
Discover Bladder Cancer Investors, Funding Rounds, Invested Amounts, and Funding Growth
Bladder Cancer News
Stay informed and ahead of the curve with TrendFeedr’s News feature, which provides access to 5.9K Bladder Cancer articles. The tool is tailored for professionals seeking to understand the historical trajectory and current momentum of changing market trends.
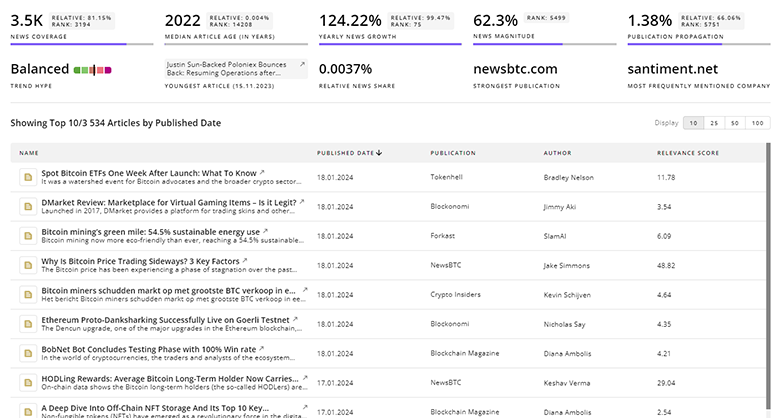
5.9K Bladder Cancer News Articles
Discover Latest Bladder Cancer Articles, News Magnitude, Publication Propagation, Yearly Growth, and Strongest Publications
Executive Summary
The strategic inflection in bladder cancer is straightforward: control of the diagnostic gate—accurate, reimbursed, non-invasive urine tests—determines which therapies become commercially viable at scale. Companies that pair high-sensitivity diagnostics with a clear therapeutic pathway (either a validated intravesical delivery system or a targeted immunotherapy) will extract the most value. Health systems will reward solutions that demonstrably reduce lifetime surveillance costs and avoid avoidable cystoscopies without sacrificing oncologic safety. For investors and operators, the practical playbook is to (1) secure validated diagnostic signatures with reimbursement evidence, (2) couple diagnostics to a near-term therapeutic pathway that reduces hospital throughput (sustained-release intravesical agents or outpatient procedural tools), and (3) build the real-world data infrastructure that enables outcome-based contracting.
Interested in enhancing our coverage of trends and tech? We value insights from experts like you - reach out!