Drug Development Report
: Analysis on the Market, Trends, and TechnologiesThe drug-development landscape is compressing risk through data and specialization: industry funding for the topic totals $54.40B, concentrated across 1,572 active companies, creating intense competition for execution capability. While 66% of large firms now use AI across recruitment, analytics and real-world data—raising potential ROI by up to 2.2× when governance is clear PPD Pulse Report – Global R&D Insights in Pharmaceuticals, 2024—nearly 40% of organizations report rising trial complexity and 45% report longer development timelines than two years ago. These pressures favor companies that combine predictive modeling, model-informed development and specialized manufacturing to shorten cycle time and reduce late-stage attrition.
We updated this report 120 days ago. Noticed something’s off? Let’s make it right together — reach out!
Topic Dominance Index of Drug Development
The Topic Dominance Index trendline combines the share of voice distributions of Drug Development from 3 data sources: published articles, founded companies, and global search
Key Activities and Applications
- AI-enabled target and candidate triage — applying deep learning and generative models to prioritize targets and design molecules reduces the experimental burden in hit-to-lead phases; in silico approaches now compete with physical HTS for early signal generation.
- Model-Informed Drug Development (MIDD) for dose and design optimization — mechanistic PK/PD and QSP models inform First-in-Human dose choices and regulatory interactions, materially reducing uncertainty at IND and beyond RES Group, Inc.
- Preclinical human-relevant testing and animal-free batteries — organoids, human cell assays and AI-driven bioanalytics replace or augment animal studies to improve translatability and satisfy evolving regulatory expectations Predictability of preclinical development.
- Adaptive, decentralized and hybrid clinical execution — patient-centric designs and digital monitoring reduce enrollment friction and manage protocol complexity that now limits many programs.
- Specialized CDMO/CDMO+ services for complex modalities — biologics, ADCs, oligonucleotides and long-acting injectables require integrated substance/product capabilities and high-containment facilities; CDMOs with these competencies capture premium demand.
Emergent Trends and Core Insights
- AI adoption moves into operational risk management — firms that apply ML to trial risk, patient selection and translational prediction secure concrete cost and time benefits; adoption is broadening beyond discovery to program management Intelligencia AI.
- Escalating trial complexity reshapes value capture — with ~40% of companies flagging protocol intricacy, demand rises for adaptive designs, digital biomarkers and AI-driven monitoring to avoid timeline erosion.
- Therapeutic and modality concentration — oncology, immunology and anti-diabetics make up >70% of innovative biologic market value; ADCs, GLP-1s and mRNA-derived vaccines dominate near-term pipeline value IQVIA Early Bird.
- Computational discovery accelerates service-market growth — computational/in-silico discovery is forecast to expand at a high CAGR, driving platform and software services growth to USD 197.0B by 2029 at ~12% CAGR Drug Discovery Technologies – BCC Research.
- Regulatory and payer pressure on pricing increases focus on evidence generation — stronger expectations for RWD/RWE and cost-effectiveness evidence push developers to front-load data capture and align trials to reimbursement requirements Frost & Sullivan – Global Pharmaceutical Industry Outlook.
Technologies and Methodologies
- Generative chemistry and AI-driven molecular design — generative models and reinforcement learning for lead generation reduce time-to-lead and enable exploration of non-intuitive chemical space.
- Mechanistic QSP and PBPK modeling suites — cloud-enabled simulation platforms (MonolixSuite, BIOiSIM and others) make MIDD operational across preclinical and clinical stages, improving dosing and cohort selection.
- Digital twins and biosimulation for translational fidelity — disease-specific virtual models enable scenario testing and reduce failed translational bets, limiting costly in-vivo iterations.
- High-content phenotypic and single-cell screens coupled with AI analytics — phenotypic HCS and single-cell CRISPR screens produce multi-modal readouts that feed ML pipelines for improved hit prioritization Pharmaron.
Key operational inference: organizations that combine predictive modeling with executional sovereignty (manufacturing + formulation + clinical operations) capture margin and accelerate go/no-go decisions, while players that provide single-function services face pricing pressure unless they hold unique IP.
Drug Development Funding
A total of 2.5K Drug Development companies have received funding.
Overall, Drug Development companies have raised $293.9B.
Companies within the Drug Development domain have secured capital from 11.0K funding rounds.
The chart shows the funding trendline of Drug Development companies over the last 5 years
Drug Development Companies
- VeriSIM Life — VeriSIM Life builds AI-enabled biosimulation disease models to improve translational prediction and reduce reliance on animal studies; their platform aims to accelerate candidate triage and produce stronger preclinical evidence packages for INDs, addressing the high attrition of animal-based screening.
- ForDoz Pharma Corp — ForDoz Pharma Corp is a U.S. sterile-injectable CDMO focused on complex formulations including liposomal, nanoparticle and in-situ depot long-acting injectables; by concentrating on sterile manufacturing capacity in the United States they aim to solve supply and scale constraints for advanced injectables.
- DNTOX GmbH — DNTOX GmbH offers human cell-based, AI-paired bioanalytical testing batteries for developmental neurotoxicity, targeting chemical and lead-safety screening without animals; their approach aligns with regulatory movement toward human-relevant NAMs and supports safer lead selection earlier in development.
- PhinC Development — PhinC Development specializes in PK/PD modeling and Model-Informed Drug Development services for small and mid-sized sponsors; their proficiency in PBPK and population modeling reduces dose-finding risk and supports regulator-facing evidence packages that compress clinical timelines.
- PROSION Therapeutics — PROSION Therapeutics pursues chemical building-block platform technology to address historically undruggable oncology targets; with a modular, block-assembly chemistry approach they aim to generate differentiated molecules for difficult targets while keeping preclinical optimization lean.
Gain a better understanding of 7.5K companies that drive Drug Development, how mature and well-funded these companies are.
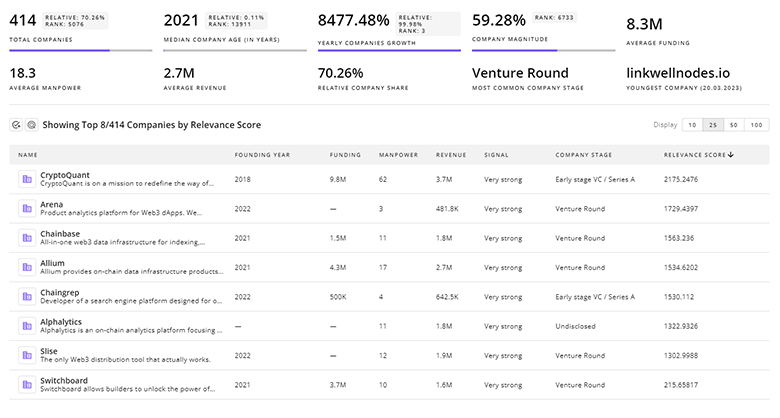
7.5K Drug Development Companies
Discover Drug Development Companies, their Funding, Manpower, Revenues, Stages, and much more
Drug Development Investors
Gain insights into 8.7K Drug Development investors and investment deals. TrendFeedr’s investors tool presents an overview of investment trends and activities, helping create better investment strategies and partnerships.
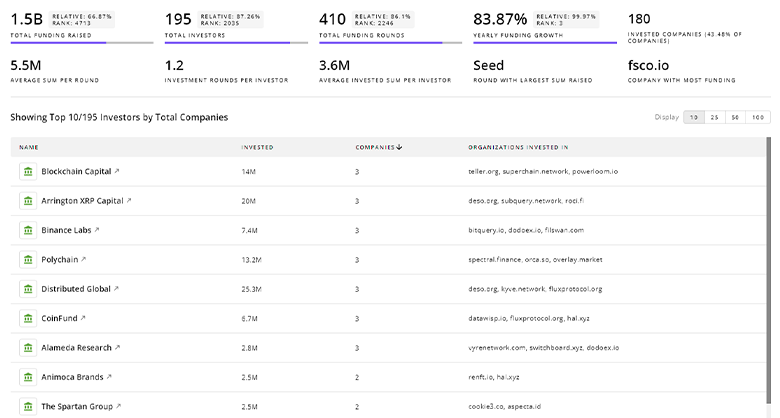
8.7K Drug Development Investors
Discover Drug Development Investors, Funding Rounds, Invested Amounts, and Funding Growth
Drug Development News
Gain a competitive advantage with access to 25.4K Drug Development articles with TrendFeedr's News feature. The tool offers an extensive database of articles covering recent trends and past events in Drug Development. This enables innovators and market leaders to make well-informed fact-based decisions.
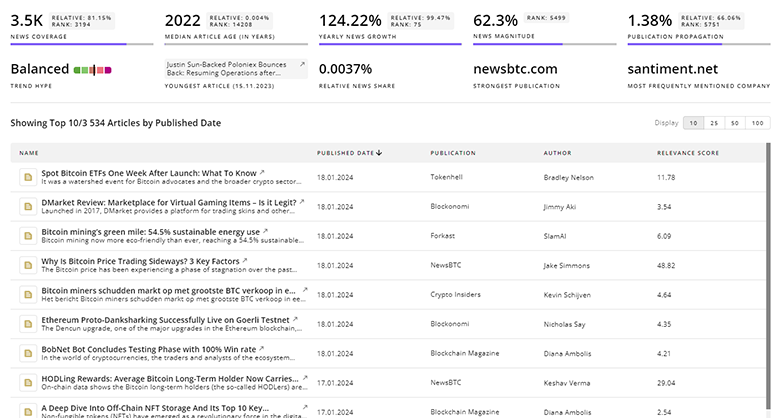
25.4K Drug Development News Articles
Discover Latest Drug Development Articles, News Magnitude, Publication Propagation, Yearly Growth, and Strongest Publications
Executive Summary
Drug development is now a two-track contest: predictive intelligence that reduces translational risk and specialized execution capability that delivers complex modalities at scale. Financial flows and patent activity confirm that informatics, biosimulation and MIDD are no longer academic experiments but core strategic assets, while CDMOs and integrated CROs that offer high-containment manufacturing, advanced formulation IP and rapid clinical supply unlock near-term commercial value. Sponsors should therefore allocate capital to secure both a validated predictive layer for go/no-go decisions and geographically diversified execution partners for biologics and advanced delivery systems; failing to do so increases the probability of late-stage cost and time overruns given the current regulatory and payer environment.
We seek partnerships with industry experts to deliver actionable insights into trends and tech. Interested? Let us know!