Dysfunction Associated Steatohepatitis Report
: Analysis on the Market, Trends, and TechnologiesThe field of Dysfunction Associated Steatohepatitis sits at a tactical inflection where diagnostics and multifunctional therapeutics compete for short-term value capture; the topic shows 571 published articles and 118 active companies, and has attracted $27.04B in aggregate funding to date, underscoring how capital intensity has concentrated on a small set of clinical and diagnostic plays. Market research predicts rapid commercial expansion for metabolic-steatohepatitis therapies—estimates range from multi-billion-dollar current markets to double-digit CAGRs driven by pipeline approvals and screening uptake Metabolic Dysfunction-Associated Steatohepatitis Market Report. Industry activity centers on (1) demonstrating fibrosis reversal with safe systemic drugs, and (2) achieving clinical acceptance for non-invasive staging tools that reduce trial cost and accelerate patient identification; these two vectors frame short-term M&A and partnership pressure across diagnostics and therapeutics.
We updated this report 90 days ago. Noticed something’s off? Let’s make it right together — reach out!
Topic Dominance Index of Dysfunction Associated Steatohepatitis
The Topic Dominance Index trendline combines the share of voice distributions of Dysfunction Associated Steatohepatitis from 3 data sources: published articles, founded companies, and global search
Key Activities and Applications
- Non-invasive staging and triage: Development and clinical validation of blood-based panels, imaging analytics, and quantitative SPECT/MRI signatures to replace biopsy for enrollment and endpoint measurement. This activity is driven by patent flows and regulatory push to reduce procedural risk.
- Multi-target therapeutics: Design and clinical advancement of agents that simultaneously modulate metabolic drivers, inflammation, and fibrogenesis (for example, SCD-1 inhibitors, cyclophilin inhibitors, THR-β agonists). Developers aim to show histologic improvement and fibrosis regression in Phase 2/3 programs GlobalData LOA models.
- Combination regimen testing: Running trials that pair metabolic modulators (GLP-1/GIP RAs or THR-β agonists) with anti-fibrotic or immune-modulating drugs to increase responder rates, reduce heterogeneity risk, and produce stronger histologic endpoints.
- Preclinical humanized modeling for de-risking: Use of iPSC models, 3D liver spheroids and patient-derived organoids to screen on-target efficacy and chronic toxicity before expensive in vivo or clinic-scale trials.
- Real-world evidence (RWE) integration: Deploy longitudinal RWE to validate non-invasive biomarkers, demonstrate clinical utility for payers, and support label expansion once initial approvals occur.
Emergent Trends and Core Insights
- Shift from single-target failure risk to multipronged biological strategies. The pipeline now prioritizes agents addressing lipid handling, immune drive, and stellate cell activation together rather than in isolation.
- Non-invasive equivalents to biopsy are the single highest strategic choke point. Companies that establish regulatory and clinical parity for blood tests or advanced imaging will control trial enrollment economics and post-launch diagnostic flows.
- Regulatory gating is loosening for MASH endpoints but remains evidence-driven. Accelerated approvals and recent histology-based approvals have increased investor tolerance for late-stage spending, but payers demand clear RWE on clinical outcomes and fibrosis improvement.
- Geographic concentration of commercial value in high-income markets. North America remains the primary launch market and revenue driver; Asia-Pacific shows the fastest patient prevalence growth and thus an essential secondary commercial target.
- Microbiome and gut-liver axis therapeutics are maturing from hypothesis to IP commitments. Recent patent filings and early translational studies indicate growing commercial bets on microbiome modulation as a treatment modality.
Technologies and Methodologies
- Advanced blood-based biomarker panels combining proteomics, lipidomics, and metabolomics to detect lobular inflammation and ballooning without histology; patents and prototype assays target multi-analyte signatures over simple enzyme ratios.
- Quantitative imaging and nuclear function testing, including liver SPECT indices and MRI-PDFF/elastography integration, to provide objective, insurance-reimbursable liver function and steatosis metrics Echosens FibroScan®.
- iPSC and 3D spheroid platforms for translational screening and chronic toxicity studies that emulate human responses, improving preclinical selection and reducing late-stage attrition DefiniGEN.
- Small-molecule modulation of metabolic master regulators (for example, SREBP and THR-β targeting) to reduce hepatic lipid accumulation and downstream fibrogenic signaling GlobalData LOA for THR-β agents.
- AI/ML image and EHR analytics for progression risk scoring, trial enrichment, and automated elastography interpretation to standardize endpoints across sites Precision MedCare ultrasound ML application.
Dysfunction Associated Steatohepatitis Funding
A total of 60 Dysfunction Associated Steatohepatitis companies have received funding.
Overall, Dysfunction Associated Steatohepatitis companies have raised $27.0B.
Companies within the Dysfunction Associated Steatohepatitis domain have secured capital from 381 funding rounds.
The chart shows the funding trendline of Dysfunction Associated Steatohepatitis companies over the last 5 years
Dysfunction Associated Steatohepatitis Companies
- Metadeq Diagnostics — Metadeq develops HeparDx, a blood test positioned to detect lobular inflammation and hepatocyte ballooning for MASH and fibrosis staging. The company emphasizes early detection and monitoring pathways that align with recent guideline recommendations for universal screening in high-risk patients. Its small team focuses on clinical validation to secure payor coverage and trial integration. Metadeq is an acquisitive target for therapeutics firms seeking an on-ramp to patient identification.
- AGED Diagnostics — AGED Diagnostics applies transcriptional and spatial transcriptomics to generate multi-gene signatures for early liver disease and tumorigenesis detection. Despite modest funding, the firm has attracted institutional validation for its molecular approach and aims to position its assay as a higher-sensitivity triage for imaging and specialist referral. The company’s technical differentiation lies in tissue-level spatial resolution applied to blood-derived signals.
- HepaPredict AB — HepaPredict operates a 3D primary human liver spheroid CRO platform spun out of an academic center to offer preclinical metabolism, toxicity, and chronic response data. The platform is optimized for co-culture with non-parenchymal cells and supports chronic dosing studies that conventional in vitro models cannot reproduce. This capability reduces translational risk for drug developers and creates a specialized service play with high strategic value to mid-stage biotechs.
- Servatrix Biomed — Servatrix is a university spin-off developing NRF2 modulators that aim to modulate oxidative and metabolic stress in MASH. Preclinical data show inhibition of steatosis accumulation and attenuation of fibrosis progression without excessive NRF2 activation. The firm’s approach targets cellular resilience and could complement metabolic agents or serve as a combination partner in trials focused on fibrosis endpoints.
- SJT Molecular Research — SJT Molecular discovered a family of orally bioavailable small molecules that target multiple metabolic syndrome components relevant to MASH (obesity, steatosis, dyslipidemia). The company pursues a low-cost oral monotherapy strategy that may be attractive in regions with limited access to injectables. With a compact team and patented chemistry, SJT represents a pragmatic therapeutic entrant that could compete on pricing and access if clinical efficacy is shown.
Gain a better understanding of 118 companies that drive Dysfunction Associated Steatohepatitis, how mature and well-funded these companies are.
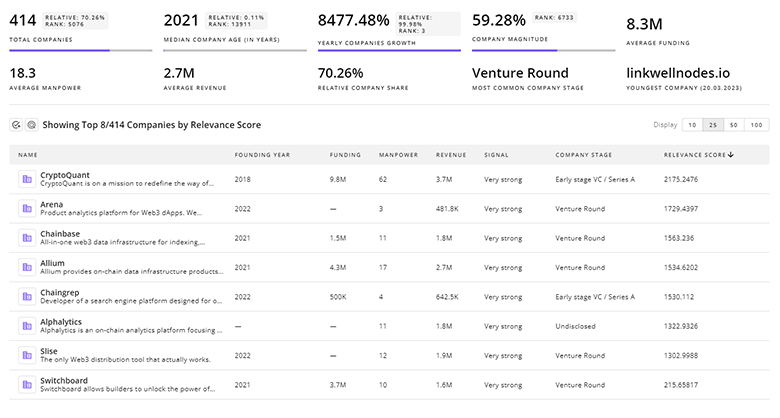
118 Dysfunction Associated Steatohepatitis Companies
Discover Dysfunction Associated Steatohepatitis Companies, their Funding, Manpower, Revenues, Stages, and much more
Dysfunction Associated Steatohepatitis Investors
Gain insights into 492 Dysfunction Associated Steatohepatitis investors and investment deals. TrendFeedr’s investors tool presents an overview of investment trends and activities, helping create better investment strategies and partnerships.
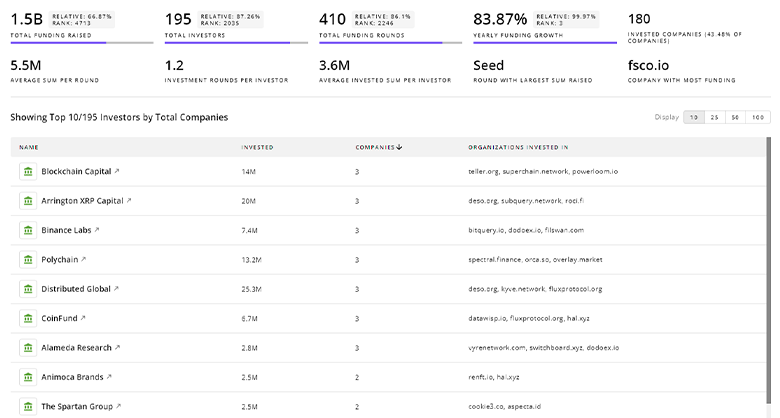
492 Dysfunction Associated Steatohepatitis Investors
Discover Dysfunction Associated Steatohepatitis Investors, Funding Rounds, Invested Amounts, and Funding Growth
Dysfunction Associated Steatohepatitis News
Gain a competitive advantage with access to 571 Dysfunction Associated Steatohepatitis articles with TrendFeedr's News feature. The tool offers an extensive database of articles covering recent trends and past events in Dysfunction Associated Steatohepatitis. This enables innovators and market leaders to make well-informed fact-based decisions.
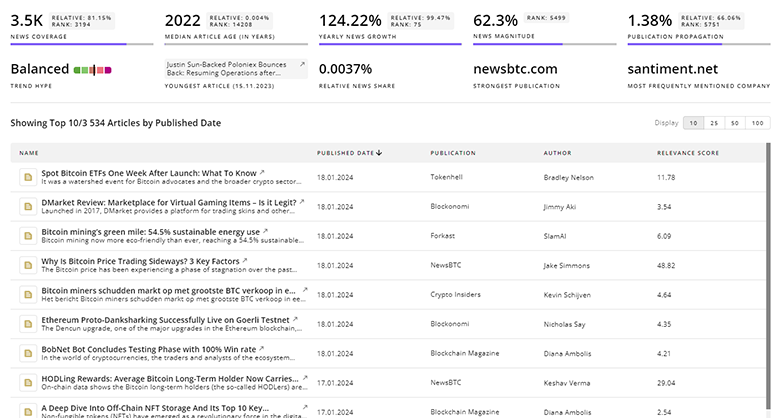
571 Dysfunction Associated Steatohepatitis News Articles
Discover Latest Dysfunction Associated Steatohepatitis Articles, News Magnitude, Publication Propagation, Yearly Growth, and Strongest Publications
Executive Summary
The business logic for Dysfunction Associated Steatohepatitis is straightforward: clinical and commercial value will concentrate on those technologies that either (a) make trials cheaper and faster by providing validated non-invasive staging and enrolment tools, or (b) deliver reproducible fibrosis regression with safety profiles acceptable to regulators and payers. Firms that combine a scalable diagnostic ingredient with clear utility in trial enrichment or market launch will command premium acquisition terms; conversely, therapeutic developers face a will-pay test where payer adoption will depend on demonstrable clinical outcomes and RWE. For investors and strategic teams, priority actions are (1) secure partnerships between diagnostic platform providers and therapy sponsors to embed staging tools into pivotal studies, and (2) invest in preclinical humanized modeling or CRO capabilities that materially reduce Phase 2→3 attrition risk. The capital already deployed ($27.04B) and the high patent activity around non-invasive diagnostics and metabolic targets create a narrow window in which focused commercial and clinical execution will determine the winners.
We seek partnerships with industry experts to deliver actionable insights into trends and tech. Interested? Let us know!