
Femtech Report
: Analysis on the Market, Trends, and TechnologiesThe femtech sector is at a decisive inflection where capital now rewards measurable clinical outcomes over attention metrics—median funding per company is $3.15M even as aggregate capital deployed exceeds $16.0B—creating a premium for products that convert proprietary physiological data into validated clinical benefit. Market sizing is uneven across studies but shows a clear pattern: device-heavy segments and pregnancy/clinic-oriented end markets capture the largest revenue pools, while AI and high-throughput laboratory technologies are driving the next wave of clinical evidence generation.
This article was last updated 86 days ago. If you find any info is missing, let us know!
Topic Dominance Index of Femtech
The Dominance Index of Femtech looks at the evolution of the sector through a combination of multiple data sources. We analyze the distribution of news articles that mention Femtech, the timeline of newly founded companies working in this sector, and the share of voice within the global search data
Key Activities and Applications
- Precision reproductive diagnostics — Automated image and data analytics to stratify embryo and oocyte quality, improving IVF decisioning and lowering cycle failure rates; exemplified by objective AI image analysis tools that report ~90% fertilization prediction accuracy in clinical validations.
- Continuous fertility and vaginal-health monitoring — Internal daily sampling devices that measure cervical mucus and vaginal biomarkers to define full fertile windows and pelvic-floor state, enabling office-connected therapeutic pathways. kegg has supported 60,000 users to date.
- At-home bioelectronic therapeutics for pelvic disorders — Wearable or self-placed neuromodulation and mechanotherapy devices that aim to deliver surgical-level symptom reduction without implants or OR time. These address stress urinary incontinence and overactive bladder as first commercial use cases. Flyte by Pelvital
- Minimally invasive structural interventions — Office or short-procedure systems to treat fibroids and pelvic organ prolapse with fewer complications and faster recovery compared with historical surgical options. Regulatory clearances and post-market studies are central to adoption. FEMSelect
- Deep-tech enabling platforms for R&D and manufacturing — High-precision microfabrication, droplet-scale microfluidics and non-visual defect inspection services that reduce device time-to-prototype and feed AI training datasets for diagnostics and biologics. These platform plays act as industrial enablers for clinical-grade products.
Emergent Trends and Core Insights
- Devices are the dominant revenue engine as clinical-grade wearables and diagnostic hardware secure the largest share of product revenue; investors are shifting allocation toward capital-intensive, evidence-oriented device companies RootsAnalysis – Femtech Market.
- Data-to-therapy pipelines create defensibility: organizations collecting proprietary internal physiological signals (vaginal, cervical, embryology images, microfluidic screening outputs) can monetize both clinical decision support and downstream therapeutics when they attach clinical outcome evidence. Proprietary biosignal capture is now a strategic moat.
- AI as a clinical adjudicator, not a marketing feature: AI models that demonstrate reproducible improvements in clinical endpoints (implantation rates, clinic workflow reduction, measurable ROI for employers/health systems) command premium exit and partnership terms.
- Geographic concentration with shifting growth pockets: North America accounts for the largest share of revenue and funding, while Asia-Pacific is the fastest-growing region driven by smartphone penetration and telehealth expansion; regulatory harmonization in US/EU eases device commercialization in those markets GM Insights – Femtech Market.
- Outcome-first commercialization: payers and large employers increasingly require demonstrable ROI (reduced NICU admissions, lowered surgical rates) before contracting femtech services, accelerating consolidation and strategic partnerships with healthcare delivery systems.
Technologies and Methodologies
- AI/ML for objective clinical scoring — Single-image and time-lapse embryo assessment, ovulation/hormone state modeling, and symptom-to-risk stratification models that integrate multi-modal inputs (image, wearable biometrics, self-reported outcomes). These systems are being validated in clinic networks and regulatory submissions MIM Fertility.
- Sensor fusion wearables and vaginal biosensors — Multi-parameter fusion (skin/aural temperature, HRV, electrodermal signals, vaginal mucus sensors) used to infer hormonal states and early warning for complications; successful products pair continuous sensing with clinician workflows.
- Microfluidics and droplet-scale screening — High-throughput, ultra-low volume droplet handling enabling rapid biological library synthesis and screening that produces the large, labeled datasets needed for predictive models in reproductive biology and drug discovery.
- Femtosecond laser microfabrication and wafer-level glass processing — Enables complex micro-optical and microfluidic components at scale for implantable sensors and diagnostic cartridges FEMTOPRINT SA.
- Remote patient monitoring (RPM) + clinical integration — RPM protocols that export validated device data into EHRs and clinical decision pathways, enabling reimbursement pathways and tighter hospital/clinic partnerships.
Femtech Funding
A total of 941 Femtech companies have received funding.
Overall, Femtech companies have raised $14.6B.
Companies within the Femtech domain have secured capital from 3.2K funding rounds.
The chart shows the funding trendline of Femtech companies over the last 5 years
Femtech Companies
- Lady Technologies — kegg positions the company at the intersection of vaginal biosensing and fertility guidance by measuring cervical mucus changes to map the full fertile window; the device’s daily in-vagina use and the company’s ~60,000 user base create a unique longitudinal dataset for parity with clinic workflows. Company financials show $9.50M raised and a 36-person team focused on scaling clinical integrations.
- FemPulse Corporation — Developing a small, patient-self-placed wearable neuromodulation device for bladder dysfunction that avoids surgery; current clinical evaluations emphasize at-home control via smartphone and the potential to broaden access to neuromodulation without implants. Total capital raised: $13.50M</strong>; targeted indication: Overactive Bladder.
- Femiguard — Offers a reversible, non-hormonal fallopian-ostium occlusion device aimed at preventing hydrosalpinx fluid spill and improving IVF implantation success; the anatomical device targets a high-impact clinical niche and emphasizes measurable downstream IVF outcome gains. Early capital is modest ($900K) and the team size is intentionally small to accelerate focused clinical adoption.
- Future Fertility — Commercializes AI image-analysis tools (VIOLET™ & MAGENTA™) for objective oocyte assessment that report ~90% fertilization-prediction accuracy; deployed across 180 clinics in 32 countries, the product converts lab microscopy into immediate clinical decision support, shortening time-to-answer in IVF workflows. Raised: $6.0M.
- FemtoFluidics — Platform developer for droplet-scale liquid handling that moves 1,000x smaller droplets at 1,000x speed vs legacy robotics, producing orders-of-magnitude more experimental throughput; the company positions itself as the data engine for AI-driven reproductive and drug discovery pipelines rather than a direct consumer femtech product. Early venture support totals $680K.
Uncover actionable market insights on 6.2K companies driving Femtech with TrendFeedr's Companies tool.
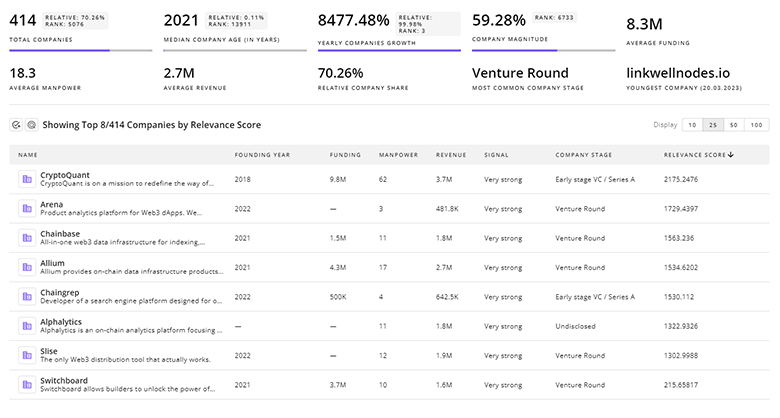
6.2K Femtech Companies
Discover Femtech Companies, their Funding, Manpower, Revenues, Stages, and much more
Femtech Investors
Get ahead with your investment strategy with insights into 3.1K Femtech investors. TrendFeedr’s investors tool is your go-to source for comprehensive analysis of investment activities and financial trends. The tool is tailored for navigating the investment world, offering insights for successful market positioning and partnerships within Femtech.
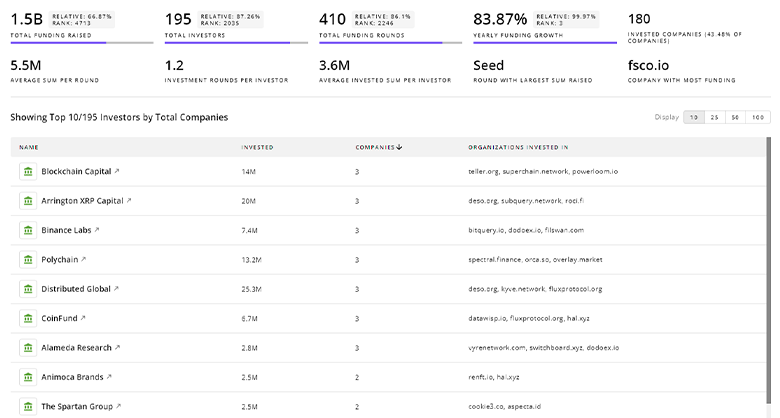
3.1K Femtech Investors
Discover Femtech Investors, Funding Rounds, Invested Amounts, and Funding Growth
Femtech News
TrendFeedr’s News feature offers access to 4.9K news articles on Femtech. The tool provides up-to-date news on trends, technologies, and companies, enabling effective trend and sentiment tracking.
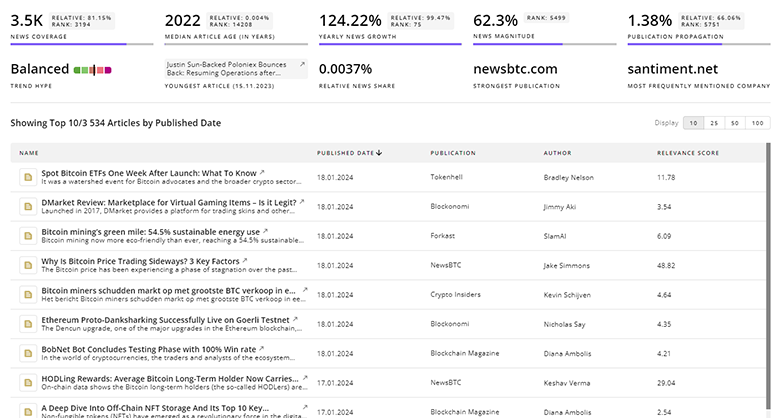
4.9K Femtech News Articles
Discover Latest Femtech Articles, News Magnitude, Publication Propagation, Yearly Growth, and Strongest Publications
Executive Summary
Femtech is shifting from attention and community plays toward rigorous, clinically anchored innovation. Investors, health systems, and procurement teams now prize measurable outcome improvements—reduced neonatal complications, demonstrable IVF success lift, and clinic workflow efficiencies—over raw user counts. The companies most likely to capture value will combine proprietary biosignal capture, validated AI decision support, and regulatory-grade device development while integrating into clinician workflows. Strategic priorities for stakeholders should be: fund and scale product clinical studies that tie features directly to reduced care cost or improved outcomes; invest in manufacturing and microfabrication partners to control quality and supply; and merge outcome data with reimbursement conversations early to accelerate enterprise adoption.
Have expertise in trends or technology? Your input can enrich our content — consider collaborating with us!











