Pharmaceutical Manufacturing Report
: Analysis on the Market, Trends, and TechnologiesThe pharmaceutical manufacturing sector is in a rapid, capital-intensive transition: legacy batch volumes sit alongside a rising need for continuous, sensor-rich production and sterile fill-finish capacity, and market data places the industry size at $525,000,000,000 in 2024—a baseline that underpins investment decisions today. Investment flows and forecasts from independent market analysts further show mid-to-high single-digit CAGRs into the next decade, concentrating value in injectables, biologics, and advanced CDMO services Pharmaceutical Manufacturing Market Size, Share & Growth.
The last time we updated this report was 89 days ago. If there’s something missing or off, your tips are welcome!
Topic Dominance Index of Pharmaceutical Manufacturing
The Dominance Index for Pharmaceutical Manufacturing merges timelines of published articles, newly founded companies, and global search data to provide a comprehensive perspective into the topic.
Key Activities and Applications
- Contract development and manufacturing (CDMO) services for sterile injectables and complex small molecules, enabling sponsors to access specialized aseptic processing and lyophilization capacity.
- Active Pharmaceutical Ingredient (API) synthesis and intermediates production, including high-potency and niche chemistries that require containment and specialized downstream processing.
- Sterile fill-finish and lyophilization for biologics, viral vectors and complex parenterals; this activity includes pre-filled syringes, cartridges and on-body delivery integration.
- Continuous flow and continuous bioprocessing operations for both API and finished dosage forms to reduce cycle time, lower inventory and improve quality attributes Global Pharmaceutical Continuous Manufacturing Market Growth 2025, Forecast To 2034.
- Advanced packaging and serialization with attention to extractables, compatibility and supply-chain traceability for regulated markets.
- Analytical and QC services (PAT and real-time monitoring) to support real-time release testing and shorter release cycles.
Emergent Trends and Core Insights
- Shift to continuous and hybrid production: Industry forecasts highlight accelerating adoption of continuous manufacturing for both APIs and finished doses; this is creating an equipment and validation market growing at double-digit rates in many analyses Pharmaceutical Continuous Manufacturing Market Size, Growth, Analysis, 2034.
- Sterile capacity tightness: Demand for fill-finish slots (especially for biologics, viral vectors and ADCs) is outpacing greenfield capacity additions, raising time-to-market risk for sponsors that cannot secure qualified CDMO partners.
- Geographic rebalancing and policy incentives: Governments are deploying fiscal measures (approx. $20,000,000,000 in incentives cited across reports) to onshore or near-shore APIs and critical drug production, shifting deal flows and accelerating regional investment decisions Global Pharmaceutical Manufacturing Market Outlook to 2028.
- Digital control and the MES/PAT stack as differentiators: Companies that tie real-time analytics, digital twins and validated MES layers into manufacturing capture measurable reductions in defect rates and validation time—this is now a gating capability for premium CDMO contracts Manufacturing simulation: Optimizing pharmaceutical plant design for smart production.
- Packaging moves from commodity to technical gate: Primary packaging suppliers that demonstrate low extractables, PFAS-free materials and regulatory documentation broaden addressable market, especially for sensitive biologics SGD Pharma.
- Talent and standards gap: Rapid technology adoption is exposing shortages in qualified bioprocess engineers and harmonized validation standards, which lengthens commercial scale-up timelines.
Technologies and Methodologies
- Continuous flow reactors and continuous downstream platforms for API and FDF manufacturing, reducing inventory and enabling smaller effective lot sizes.
- Single-use/disposable bioprocessing systems for rapid changeover in biologics and viral vector production, lowering cross-contamination risk and shortening campaign setup.
- Digital twin simulations and PAT integration used during design, validation and RTRT (real-time release testing) to accelerate regulatory acceptance and reduce batch release time.
- Advanced aseptic robotics and isolator systems for high-volume fill-finish of parenterals, reducing human touchpoints and contamination vectors.
- High-containment HPAPI suites and isolator lines for oncology payloads and ADC intermediates, combining containment engineering with specialized material handling.
- 3D printing and additive manufacturing for complex solid dosage forms, enabling tailored release profiles and rapid prototyping for niche products.
Pharmaceutical Manufacturing Funding
A total of 1.1K Pharmaceutical Manufacturing companies have received funding.
Overall, Pharmaceutical Manufacturing companies have raised $154.5B.
Companies within the Pharmaceutical Manufacturing domain have secured capital from 3.7K funding rounds.
The chart shows the funding trendline of Pharmaceutical Manufacturing companies over the last 5 years
Pharmaceutical Manufacturing Companies
- Pharmatory Ltd — Pharmatory is a small cGMP custom synthesis and small-scale API provider that focuses on process R&D, scale-up and high-quality cGMP manufacture for early clinical supply. Its value proposition is tight process development for challenging intermediates and fast analytical turnaround, making it a practical partner for sponsors prioritizing speed to clinic. Pharmatory positions itself for projects where large CDMOs are over-scaled or inflexible.
- Pharmatechprocess — Pharmatechprocess supplies custom, turnkey liquid processing plants and process equipment (reactors, CIP/SIP, sterile heat exchangers) and couples engineering delivery with CQV services. Their model reduces project coordination risk for mid-sized biologics and sterile drug product launches by delivering validated skids and execution support.
- 3P innovation — 3P innovation builds bespoke automation and robotic fill-finish cells specifically for aseptic processing and high-precision liquid dosing. The company's engineering focus shortens qualification timelines for CDMOs adopting automated aseptic lines and supports rapid changeovers for small-batch biotherapeutics.
- Pharmadule Morimatsu AB — Pharmadule pioneered modular facility delivery for pharma manufacturing and offers prefabricated cleanroom modules and process super-skids that compress construction risk and commissioning time. Their turnkey modular approach is attractive to sponsors seeking to deploy localized, validated capacity quickly in regulated markets.
- E-Pharma Trento — E-Pharma Trento is a CDMO specialized in solid oral delivery forms (effervescent, orally disintegrating, sublingual) and concurrently commercializes proprietary platforms it can out-license or manufacture under contract. This dual service + IP model gives customers both manufacturing capacity and platform differentiation for niche dosage forms.
Delve into the corporate landscape of Pharmaceutical Manufacturing with TrendFeedr’s Companies tool
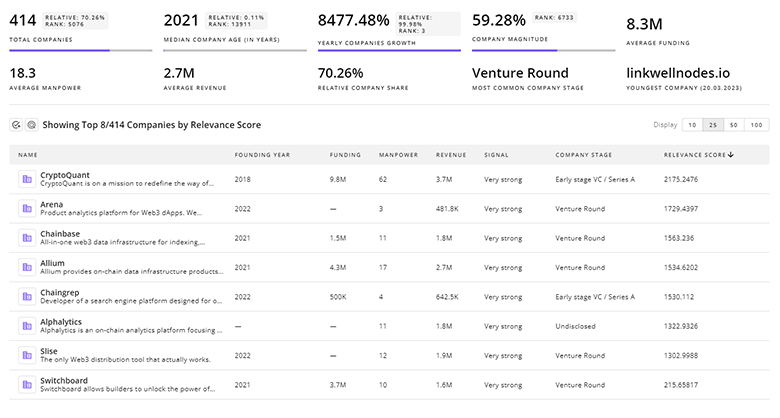
11.9K Pharmaceutical Manufacturing Companies
Discover Pharmaceutical Manufacturing Companies, their Funding, Manpower, Revenues, Stages, and much more
Pharmaceutical Manufacturing Investors
TrendFeedr’s Investors tool provides insights into 3.5K Pharmaceutical Manufacturing investors for you to keep ahead of the curve. This resource is critical for analyzing investment activities, funding trends, and market potential within the Pharmaceutical Manufacturing industry.
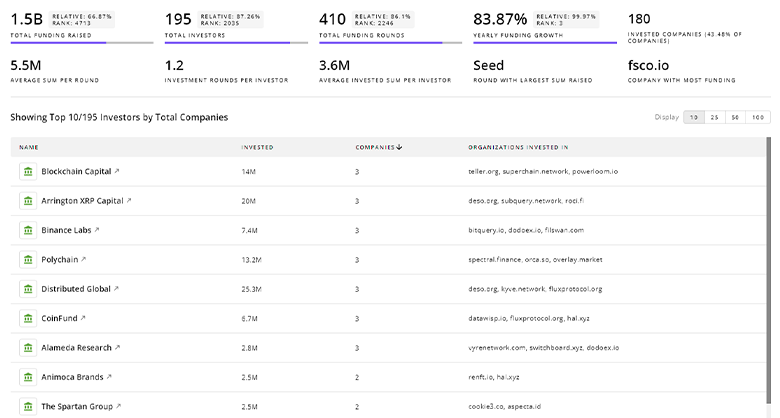
3.5K Pharmaceutical Manufacturing Investors
Discover Pharmaceutical Manufacturing Investors, Funding Rounds, Invested Amounts, and Funding Growth
Pharmaceutical Manufacturing News
TrendFeedr’s News feature offers you access to 11.8K articles on Pharmaceutical Manufacturing. Stay informed about the latest trends, technologies, and market shifts to enhance your strategic planning and decision-making.
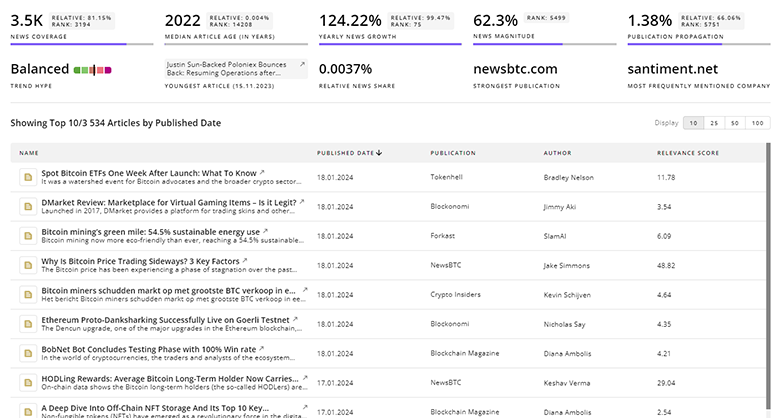
11.8K Pharmaceutical Manufacturing News Articles
Discover Latest Pharmaceutical Manufacturing Articles, News Magnitude, Publication Propagation, Yearly Growth, and Strongest Publications
Executive Summary
The pharmaceutical manufacturing landscape now separates winners by two operational choices: invest in regulatory-grade, flexible sterile and biologics capacity with a digital control backbone, or achieve extreme scale and cost advantage in commoditized generics and APIs. Sponsors and service providers must map near-term capacity needs to validate where to place multi-hundred-million dollar bets: build modular, validated assets for complex modalities or assemble optimized supply partnerships for high-volume production. In practical terms, manufacturing leaders should prioritize validated MES/PAT stacks, targeted sterile capacity investments, and transparent sustainability metrics so they can access premium commercial contracts and maintain market access in regulated jurisdictions.
Are you an insider in the trends or tech industry? We’d love for you to contribute to our content.