Pharmacovigilance Report
: Analysis on the Market, Trends, and TechnologiesThe global pharmacovigilance landscape is driven by an urgent need for comprehensive drug safety oversight, with the market projected to expand from USD 9.03 billion in 2025 to USD 20.98 billion by 2035 at an 8.8% CAGR (2025–2035) Future Market Insights. This surge is supported by a 28.14% annual growth in funding rounds within the field, underpinning 2,538 companies worldwide dedicated to adverse event monitoring and risk management. Artificial intelligence and machine learning are reshaping signal detection, while contract outsourcing now captures over 60% of market share, reflecting pharmaceutical firms’ preference for cost-efficient, scalable solutions Precedence Research.
The last time we updated this report was 264 days ago. If there’s something missing or off, your tips are welcome!
Topic Dominance Index of Pharmacovigilance
The Dominance Index for Pharmacovigilance merges timelines of published articles, newly founded companies, and global search data to provide a comprehensive perspective into the topic.
Key Activities and Applications
- Comprehensive adverse event reporting involves systematic capture and processing of individual case safety reports (ICSRs), ensuring documentation from initial notification through regulatory submission Coherent Market Insights.
- Machine learning–powered signal detection analyzes structured clinical trial data and unstructured sources for early identification of safety signals, a segment holding 39.6% of market share in 2024 Precedence Research.
- Development and execution of risk management plans integrate signal evaluation and mitigation strategies to preempt adverse outcomes IMARC Group.
- Phase IV post-marketing surveillance monitors drugs in real-world settings to assess long-term safety, accounting for 76% of market activity in 2024 Precedence Research.
- Automated literature surveillance platforms continuously scan global databases like PubMed and Embase to extract emerging safety data and regulatory updates, reducing manual review time by up to 70% CoVigilAI.
Emergent Trends and Core Insights
- Artificial intelligence and machine learning adoption in pharmacovigilance is accelerating, with the U.S. FDA issuing draft guidance on AI in drug safety in January 2025 Future Market Insights.
- Contract outsourcing now represents over 60% of the global pharmacovigilance market, driven by cost efficiency and compliance needs Precedence Research.
- Asia Pacific leads regional growth with a 10.9% CAGR through 2034, reflecting rising healthcare investment and pharmaceutical activity Precedence Research.
- North America holds approximately 33.7% of the pharmacovigilance market as of 2024, supported by stringent regulatory environments and high R&D spending IMARC Group.
- Regulatory bodies such as the EMA released AI tools and guidelines in 2024 to standardize technology use in pharmacovigilance IMARC Group.
Technologies and Methodologies
- Artificial intelligence and machine learning enhance real-time signal detection and predictive risk modeling, driving efficiency across safety workflows Future Market Insights.
- Natural language processing extracts safety insights from unstructured data sources such as clinical texts and social media posts Coherent Market Insights.
- Big data analytics platforms integrate electronic health records, claims data, and real-world evidence for comprehensive safety assessments IMARC Group.
- Cloud-based pharmacovigilance software delivers on-demand scalability, real-time monitoring, and centralized data management Allied Market Research.
- Standardized E2B(R3) XML messaging enables seamless exchange of ICSRs between sponsors and regulators, ensuring global compliance AB Cube.
Pharmacovigilance Funding
A total of 152 Pharmacovigilance companies have received funding.
Overall, Pharmacovigilance companies have raised $29.5B.
Companies within the Pharmacovigilance domain have secured capital from 536 funding rounds.
The chart shows the funding trendline of Pharmacovigilance companies over the last 5 years
Pharmacovigilance Companies
- CoVigilAI uses advanced AI algorithms to automate medical literature monitoring across Embase, PubMed, and regional journals, classifying ICSRs and conducting generative AI-based causality assessments to streamline pharmacovigilance workflows.
- Bibliovigilance offers a dedicated search engine for pharmacovigilance publications, centralizing global and national literature searches, scheduling automated queries, and maintaining detailed search histories for transparency and audit readiness.
- 3Analytics builds AI/ML/DL-driven SaaS platforms for end-to-end pharmacovigilance automation, leveraging NLP engines to harvest safety intelligence and accelerate decision-making across drug, device, and vaccine safety operations.
- NewTn SAFETY provides a cloud-native PV platform featuring real-time monitoring, customizable alerts, and DSUR/PBRER support, designed to help startups, CROs, and mid-sized pharma firms establish cost-effective safety databases.
Delve into the corporate landscape of Pharmacovigilance with TrendFeedr’s Companies tool
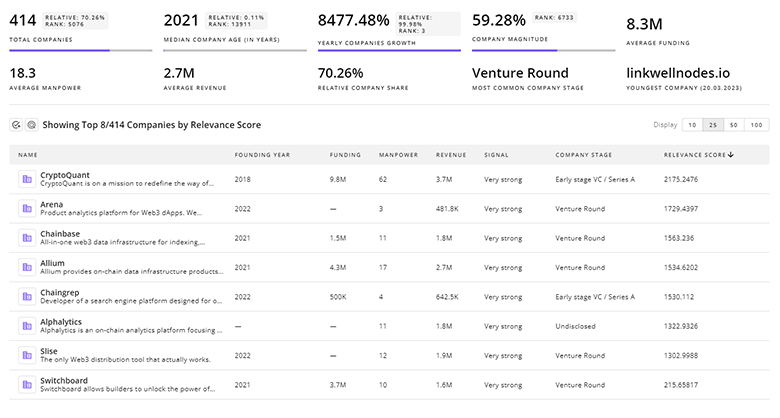
2.7K Pharmacovigilance Companies
Discover Pharmacovigilance Companies, their Funding, Manpower, Revenues, Stages, and much more
Pharmacovigilance Investors
TrendFeedr’s Investors tool provides insights into 608 Pharmacovigilance investors for you to keep ahead of the curve. This resource is critical for analyzing investment activities, funding trends, and market potential within the Pharmacovigilance industry.
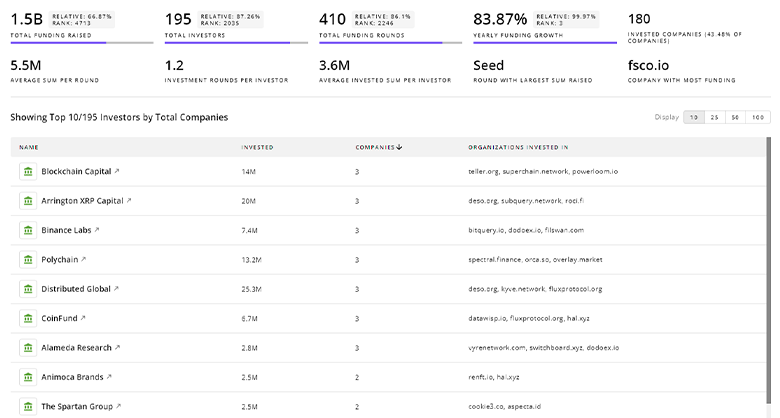
608 Pharmacovigilance Investors
Discover Pharmacovigilance Investors, Funding Rounds, Invested Amounts, and Funding Growth
Pharmacovigilance News
TrendFeedr’s News feature offers you access to 1.9K articles on Pharmacovigilance. Stay informed about the latest trends, technologies, and market shifts to enhance your strategic planning and decision-making.
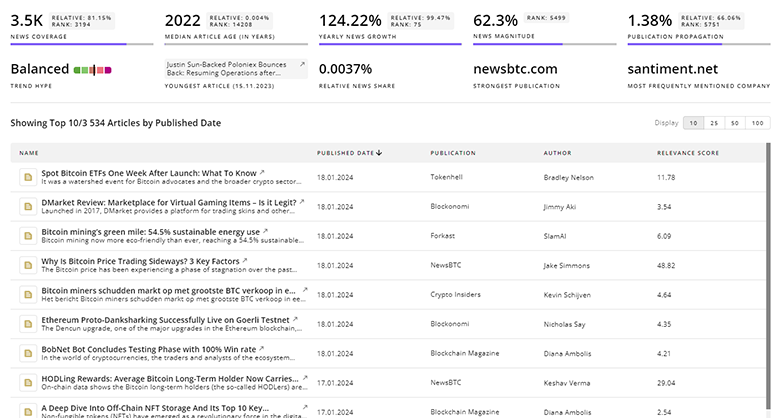
1.9K Pharmacovigilance News Articles
Discover Latest Pharmacovigilance Articles, News Magnitude, Publication Propagation, Yearly Growth, and Strongest Publications
Executive Summary
Pharmacovigilance has evolved into a high-technology, highly regulated service critical to patient safety and drug lifecycle management. Sustained market growth is propelled by rising adverse drug reactions, expanding drug pipelines, and stringent post-marketing requirements. The integration of AI, cloud computing, and advanced analytics is enhancing signal detection and risk management, while contract outsourcing remains a dominant model for scalability and cost control. Regional dynamics indicate North America’s leadership and Asia Pacific’s rapid expansion. For industry participants, success will hinge on adopting cutting-edge technologies, forging regulatory partnerships, and delivering end-to-end solutions that meet global compliance standards.
Are you an insider in the trends or tech industry? We’d love for you to contribute to our content.