Clinical Drug Development Report
: Analysis on the Market, Trends, and TechnologiesThe clinical drug development system carries heavy capital and complexity while leaving large execution gaps: $54.41B has been raised across the field and 1,571 companies operate in this space, yet patient engagement remains critically low—only 3% of U.S. physicians and patients enroll in trials and roughly ≈80% of studies miss enrollment timelines—creating a clear opportunity for targeted investments in Model-Informed Drug Development, decentralized trial infrastructure, and high-fidelity translational models to compress cycle times and reduce late-stage attrition Veeva 2025 Clinical Data Trend Report – Veeva Systems.
We updated this report 65 days ago. Noticed something’s off? Let’s make it right together — reach out!
Topic Dominance Index of Clinical Drug Development
The Topic Dominance Index trendline combines the share of voice distributions of Clinical Drug Development from 3 data sources: published articles, founded companies, and global search
Key Activities and Applications
- Model-Informed Drug Development (MIDD) and PK/PD simulation — Embedding quantitative pharmacometrics into early decision gates to reduce required patient numbers and improve dose selection; this function is central to accelerating IND to POC transitions and is actively offered by specialist partners focused on early modeling and simulation
- Decentralized and hybrid trial execution — Moving procedures and endpoints closer to patients via remote monitoring, eSource, and community access points to improve recruitment speed, retention, and diversity; industry surveys indicate DCT adoption as a direct response to chronic enrollment shortfalls
- Translational, patient-relevant preclinical testing — Deploying human-derived models (organoids, iPSC systems, spatial proteomics) and in-patient Phase 0 probes to raise confidence before Phase I, reducing the 40–50% of attrition attributable to poor translational fidelity Advanced In Vitro Models for Preclinical Drug Safety.
- Integrated CMC and platform delivery development — Creating phase-appropriate, platformized delivery solutions (autoinjectors, sustained-release, oligonucleotide processes) that link formulation, manufacturing, and clinical testing to shorten tech-transfer friction Alira Health – The 2025 Global Drug Delivery System Trends Report.
- Data-driven recruitment and site networks — Leveraging community health infrastructures (e.g. pharmacy networks) and AI for targeted outreach to resolve the persistent enrollment bottleneck that delays ~80% of trials
Emergent Trends and Core Insights
- AI and biosimulation moved to operational center stage — More than 66% of large firms report active AI use across recruitment, analytics, and RWD/RWE tasks, shifting AI from pilot projects to core trial operations and enabling automated study-build and predictive risk scoring The Pulse Report 2024 – PPD.
- Enrollment duration stabilized but remains the largest cycle-time driver — IQVIA notes a stabilization in enrollment duration in 2024 after multi-year increases; this creates an immediate tactical window to capture cycle-time gains via decentralized models and targeted recruitment tools Global Trends in R&D 2025 – IQVIA.
- Asset-centred virtualization of development — Virtual partner networks and product development teams reduce upfront overhead and allow small sponsors to buy focused program leadership and regulatory strategy rather than full-time headcount, accelerating first-in-human milestones PharmaDirections.
- Complex modalities demand integrated CDMO/CRO responses — ADCs, oligonucleotides, and cell/gene therapies push demand toward CDMOs and CROs that can co-develop CMC, analytics, and clinical delivery rather than provide point services CPC Scientific Inc..
- Regulatory pathway diversification reduces capital requirements for some programs — Pathways that allow reliance on prior data (e.g. 505(b)(2)) or model-based evidence shorten development cost and time for certain candidates, increasing the viability of repurposing and reformulation plays FDA regulatory roadmap reporting.
Technologies and Methodologies
- Generative AI and automated study-build — Use of generative models to assemble EDC/study builds and protocol drafts (examples of seven-API-call EDC automation reduce start-up time materially), increasing velocity into first patient in
- Population PK/PD and QSP/PBPK modeling — Advanced mechanistic and semi-mechanistic models underpin MIDD strategies to optimize dosing, predict exposure, and support regulatory discussions Allied Market Research biosimulation.
- Human-relevant translational assays and Phase 0 probing — Direct in-patient evaluation platforms and organoid/3D systems compress translational uncertainty and reduce late-stage failures
- Digital twins and biosimulation — Virtual trial simulations and digital twins model enrollment, endpoint sensitivity, and operational risk to select optimal designs before committing to large, costly studies.
- Integrated drug-delivery platforms — Platform autoinjectors, sustained-release technologies, and drug-device combinations serve both clinical convenience and RWE capture needs while influencing product differentiation and CMC strategy CMC Pharmaceuticals, Inc..
Clinical Drug Development Funding
A total of 529 Clinical Drug Development companies have received funding.
Overall, Clinical Drug Development companies have raised $54.4B.
Companies within the Clinical Drug Development domain have secured capital from 2.3K funding rounds.
The chart shows the funding trendline of Clinical Drug Development companies over the last 5 years
Clinical Drug Development Companies
- PhinC Development — PhinC focuses on pharmacokinetic/pharmacodynamic modeling and simulation to support early decision gates and dose selection; the firm targets small-to-mid biotech sponsors that need high-confidence, regulator-grade model outputs to shorten preclinical to clinical transitions. PhinC positions MIDD as a strategic lever to reduce trial size and accelerate go/no-go decisions, supporting sponsors through EMA/FDA expectations.
- Presage Biosciences, Inc. — Presage operates a clinical Phase 0 platform that evaluates multiple oncology agents directly in patient tumors to generate comparative efficacy signals without systemic toxicity; this method aims to refine candidate selection ahead of large trials. Their model reduces translational uncertainty by producing human-relevant, clinical-grade data packages that inform early portfolio pruning and combination selection.
- Every Cure — Every Cure builds an open-source, AI-driven repurposing engine that scores 3,000 approved drugs across 12,000 disease hypotheses to prioritize low-cost, low-risk development candidates; the nonprofit model accelerates trial readiness where safety data already exist and can leverage regulatory pathways that reduce development burden.
- RxE2 — RxE2 uses community pharmacists and AI-enabled workflows to tackle recruitment, retention, and on-site dispensing logistics, turning thousands of local pharmacies into decentralized study nodes. Their approach addresses the principal operational barrier—patient access and adherence—by integrating trusted, local care touchpoints into trial protocols.
- Celtarys Research — Celtarys supplies fluorescent chemical tools to replace radioligands in target validation and screening, enabling safer, faster, and more scalable GPCR assays; this chemical-tool specialization shortens hit-to-lead cycles and improves assay portability across discovery and translational labs.
Gain a better understanding of 1.6K companies that drive Clinical Drug Development, how mature and well-funded these companies are.
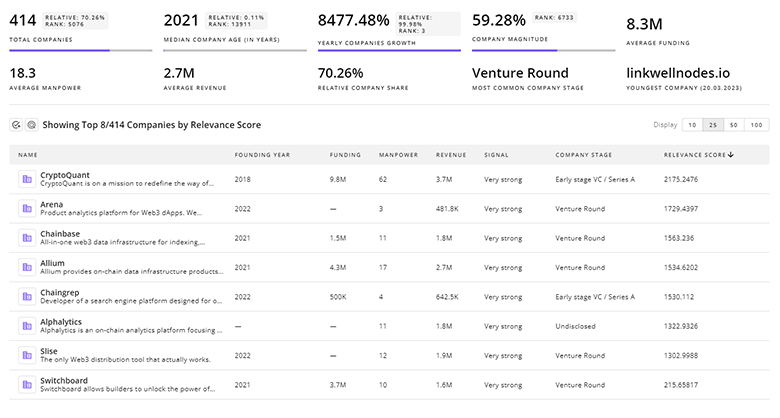
1.6K Clinical Drug Development Companies
Discover Clinical Drug Development Companies, their Funding, Manpower, Revenues, Stages, and much more
Clinical Drug Development Investors
Gain insights into 2.4K Clinical Drug Development investors and investment deals. TrendFeedr’s investors tool presents an overview of investment trends and activities, helping create better investment strategies and partnerships.
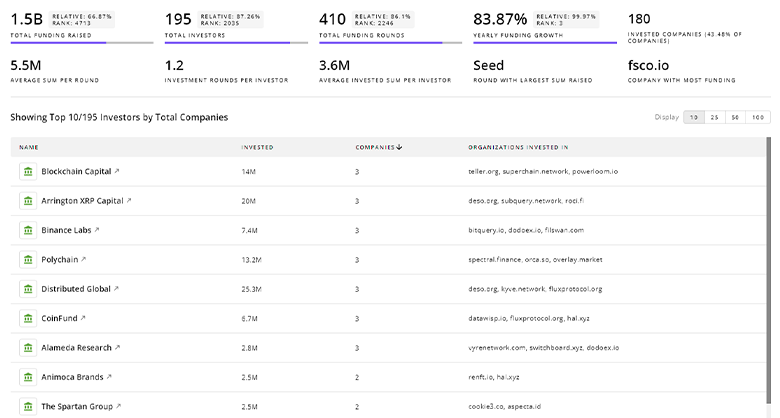
2.4K Clinical Drug Development Investors
Discover Clinical Drug Development Investors, Funding Rounds, Invested Amounts, and Funding Growth
Clinical Drug Development News
Gain a competitive advantage with access to 1.3K Clinical Drug Development articles with TrendFeedr's News feature. The tool offers an extensive database of articles covering recent trends and past events in Clinical Drug Development. This enables innovators and market leaders to make well-informed fact-based decisions.
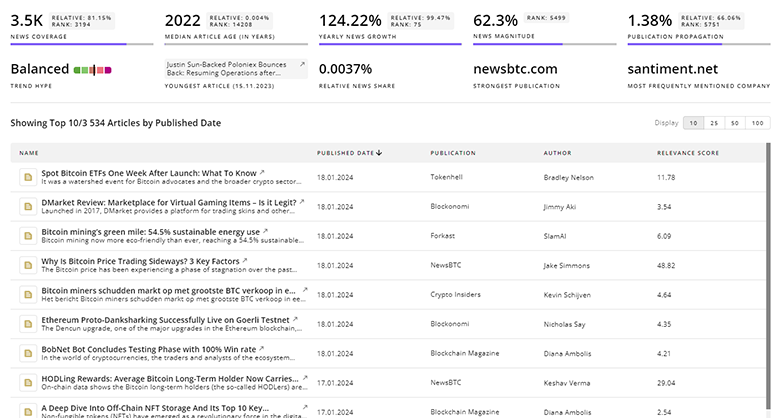
1.3K Clinical Drug Development News Articles
Discover Latest Clinical Drug Development Articles, News Magnitude, Publication Propagation, Yearly Growth, and Strongest Publications
Executive Summary
Clinical drug development now rewards organizations that combine domain expertise with predictive, data-centric approaches. Capital sits heavily in platform and AI plays, but the most immediate, high-value gains flow from fixing enrollment, improving translational fidelity, and integrating CMC with clinical execution. Sponsors should prioritize three tactical moves: embed MIDD before IND to limit patient exposure and time on trial; adopt hybrid/ decentralized recruitment channels linked to trusted community nodes; and secure translational proof points (human-relevant assays or Phase 0 probes) before committing large Phase II/III budgets. These steps reduce the dominant sources of time and value loss—slow enrollment and poor translational predictability—and create clearer, faster paths to regulatory and commercial value.
We seek partnerships with industry experts to deliver actionable insights into trends and tech. Interested? Let us know!