Nutraceutical Supplements Report
: Analysis on the Market, Trends, and TechnologiesThe nutraceutical supplements sector shows clear commercial momentum: market data records $384,800,000,000 in global size for 2024, with an internal forecast to reach $602,500,000,000 by 2030, driven by a 7.8% CAGR in the core trend dataset. Growth is concentrated in personalized nutrition, plant-derived ingredients, and higher-bioavailability delivery formats; at the same time, the industry faces elevated regulatory scrutiny and persistent adherence gaps that create both product-design and go-to-market priorities for firms that can prove clinical effect and supply-chain integrity.
We last updated this report 104 days ago. Tell us if you find something’s not quite right!
Topic Dominance Index of Nutraceutical Supplements
To gauge the impact of Nutraceutical Supplements, the Topic Dominance Index integrates time series data from three key sources: published articles, number of newly founded startups in the sector, and global search popularity.
Key Activities and Applications
Primary activity areas (market and product focus)
- Personalized nutrition and precision supplement programs: designing supplements tailored to genetic, biomarker, or lifestyle data — a major application vector for clinical-grade formulations and subscription models.
- Digestive health and gut microbiome modulation: development of probiotics, prebiotics, and postbiotic ingredients targeted at gut-brain and metabolic endpoints.
- Immune-support formulations: condition-targeted blends for immune resilience (vitamin C, zinc, botanicals) increasingly marketed for preventive health.
- Cognitive and mental-wellness stacks: nootropic combinations, adaptogens, and synbiotic approaches that aim to support cognition and stress resilience in daily life.
- Sports and performance nutrition: clinically dosed recovery, hydration, and endurance products packaged for athlete compliance and third-party certification.
- Functional food integration and nutricosmetics: embedding nutraceutical actives into foods and beauty supplements to capture routine consumption occasions Nutraceutical Market Report.
Commercial enablers and operational applications
- Contract manufacturing for rapid market entry and private label. This supports D2C brands and retailers that lack in-house production scale.
- Packaging formats and unit-dose systems that improve adherence (stick packs, sachets, blister single doses) tied to e-commerce logistics and subscription fulfillment.
- Third-party certification and lab testing to reduce trust friction and validate label claims Supplement Certified.
Emergent Trends and Core Insights
- Acceleration of personalization infrastructure: AI engines, clinician portals, and at-home testing (DNA, metabolite panels) are shifting supplementation from product-centric to plan-centric models; this trend is supported by a faster CAGR in personalized nutrition markets versus the general market.
- Ingredient sophistication outpacing raw-ingredient growth: ingredient-level forecasts show a higher CAGR than finished products, driven by plant-based isolates, standardized extracts, and novel postbiotics that can be patented and CPG-friendly Nutraceutical Ingredients Market.
- Delivery systems as a commercial wedge: liposomal, sublingual, nanoemulsion, and targeted-release particles materially change perceived efficacy and justify premium positioning; several mid-market entrants focus explicitly on these technologies SUPPLME.
- Adherence remains a structural constraint: product taste, dosing frequency, and format drive drop-off; industry reporting highlights that organoleptic barriers and regimen complexity reduce consistent usage, directing R&D toward palatable, single-serve formats and automated refill programs.
- Regulatory pressure and quality signals matter commercially: cases of adulteration and failed label claims increase demand for third-party verification and traceable supply chains; brands without verification face channel restrictions in pharmacies and healthcare recommendations Cosmeticsbusiness – Certification News.
- Distribution polarizes between D2C subscription and fortified mass channels: premium personalized players pursue subscriptions and clinician distribution while ingredient suppliers are embedding nutrients into mass-market food and beverage items for scale.
Key implication: firms that can convert clinical evidence and delivery-level IP into scalable manufacturing or platform partnerships will capture the disproportionate share of value.
Technologies and Methodologies
Core technical approaches reshaping product performance
- Liposomal and mucosal delivery: engineered phospholipid carriers and oil-suspended high-stability liposomes for rapid mucous-membrane uptake, targeting NAD+, glutathione, and fat-soluble actives Rho Nutrition.
- Microencapsulation and nano-formulations: micronization, nanoemulsions, and sustained-release coatings to protect labile actives from oxygen, moisture, and gastric degradation.
- AI-driven formulation and personalization engines: algorithms that map biomarkers, genetics, and symptom tracking to ingredient stacks and adaptive dosing rules, enabling rapid A/B testing of regimen efficacy.
- Green biomanufacturing and synthetic biology: microbial production and biosynthetic routes for melatonin, resveratrol, and other high-value extracts to secure supply and reduce ecological footprint Xtractiva Lifescience.
- Advanced extraction and standardization: supercritical CO2, dual-extraction, and cell-wall disruption for mushrooms and botanicals to produce consistent actives and standardized potency.
- Smart packaging and serialization: active barrier films, desiccants, and QR/NFC traceability for chain-of-custody and consumer engagement.
Nutraceutical Supplements Funding
A total of 863 Nutraceutical Supplements companies have received funding.
Overall, Nutraceutical Supplements companies have raised $32.9B.
Companies within the Nutraceutical Supplements domain have secured capital from 2.6K funding rounds.
The chart shows the funding trendline of Nutraceutical Supplements companies over the last 5 years
Nutraceutical Supplements Companies
- Health Protection — Health Protection operates an AI-driven personalization platform that pairs cognitive and sleep assessment tools with tailor-made nutraceutical recommendations delivered through app workflows and blood-test integration. The company’s Noobrain and Sleepie initiatives combine cognitive testing and sleep monitoring with custom supplement protocols, positioning the firm as a digital layer that prescribes product rather than only selling product. Their model targets clinicians and consumers who require evidence-linked regimens rather than one-size-fits-all packs.
- NUSEUM — NUSEUM builds an AI engine trained on thousands of clinical studies to generate precision nutrition recommendations and Food-as-Medicine integrations for partners in commerce and healthcare. The startup emphasizes enterprise PoCs with major platform partners and packages its capability as an embeddable precision-nutrition layer for retailers and insurers. Its scientific positioning and university-patented core algorithms aim to move personalized recommendations into mainstream digital channels.
- HINNAO Technology — HINNAO Technology commercializes a patented high-stability liposomal manufacturing process that suspends actives in lipid carriers for mucosal absorption, targeting fast systemic uptake of NAD+, glutathione, and other labile compounds. The company emphasizes third-party bioavailability testing and positions its delivery as a clinical-grade enhancement over traditional aqueous liposomes. By owning delivery IP, HINNAO Technology creates a defensible product layer attractive to practitioner networks and direct clinical adopters.
- BIOVIT — BIOVIT focuses on plant-derived, food-process-stable multi-nutrient ingredients and secured Innovate UK funding for a study comparing plant-derived versus synthetic nutrients. Their value proposition targets food manufacturers and large CPGs seeking to fortify processed goods with certified organic, plant-sourced nutrients that survive baking and pasteurization. This B2B ingredient strategy aims to capture scale by embedding nutraceutical value into mass consumption channels where consumer trust and regulatory traceability matter.
- NutraSky — NutraSky operates as an FDA-registered, cGMP contract manufacturer that emphasizes rapid time-to-market, low minimums, and full-service capabilities (formulation, private label, fulfillment). By combining manufacturing scale with client-facing product development support, NutraSky functions as the infrastructure backbone for D2C personalization brands that need compliant, repeatable production at speed. Their model highlights the continuing separation between data/clinical platform firms and manufacturing partners.
Enhance your understanding of market leadership and innovation patterns in your business domain.
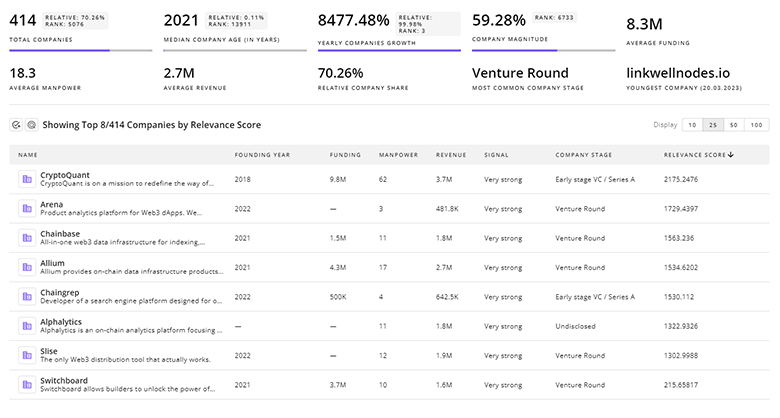
9.5K Nutraceutical Supplements Companies
Discover Nutraceutical Supplements Companies, their Funding, Manpower, Revenues, Stages, and much more
Nutraceutical Supplements Investors
TrendFeedr’s Investors tool offers comprehensive insights into 2.2K Nutraceutical Supplements investors by examining funding patterns and investment trends. This enables you to strategize effectively and identify opportunities in the Nutraceutical Supplements sector.
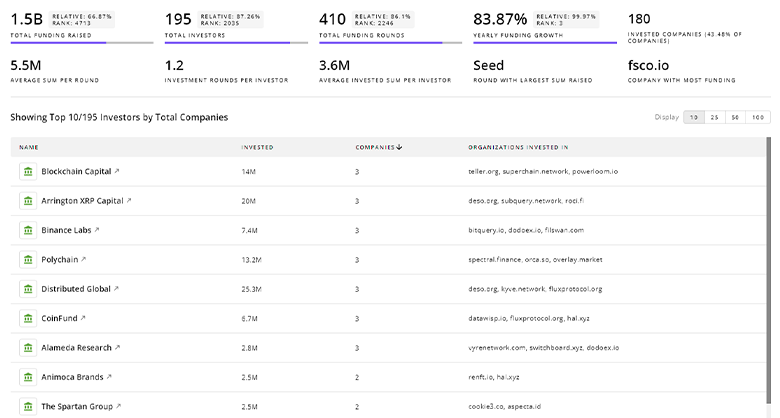
2.2K Nutraceutical Supplements Investors
Discover Nutraceutical Supplements Investors, Funding Rounds, Invested Amounts, and Funding Growth
Nutraceutical Supplements News
TrendFeedr’s News feature provides access to 3.5K Nutraceutical Supplements articles. This extensive database covers both historical and recent developments, enabling innovators and leaders to stay informed.
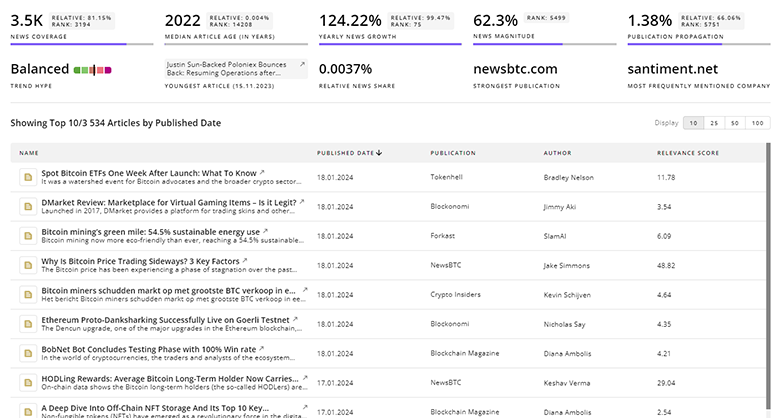
3.5K Nutraceutical Supplements News Articles
Discover Latest Nutraceutical Supplements Articles, News Magnitude, Publication Propagation, Yearly Growth, and Strongest Publications
Executive Summary
The nutraceutical supplements market is transitioning from commodity vitamin supply to a two-track industry: platformized, data-driven personalization and high-quality manufacturing/ingredient supply. Commercial winners will either secure the clinical and algorithmic moats that define what individual consumers should take, or they will dominate the certified manufacturing and delivery IP that ensures those recommendations actually work. Short-term priorities for executives and investors are clear: 1) invest in verifiable bioavailability and delivery IP to overcome adherence and efficacy skepticism; 2) lock strategic partnerships that couple personalization engines with scalable contract manufacturing; and 3) accelerate third-party verification and transparent traceability to reduce regulatory and channel risk. Firms that align clinical evidence, data platforms, and supply-chain credibility will convert high demand into defensible revenue streams.
We value collaboration with industry professionals to offer even better insights. Interested in contributing? Get in touch!