Stem Cell Biology Report
: Analysis on the Market, Trends, and TechnologiesThe stem cell biology field is entering a phase where process control and manufacturability define commercial winners rather than cell-type novelty, with the internal trend report estimating a 7.1% baseline market CAGR for core stem-cell research services and products. Market forecasts diverge—some external analyses place therapy-focused segments far higher (for example, USD 64.84 billion by 2034 at 13.27% CAGR) Stem Cell Therapy Market Size, Price, Trends | CAGR Of 13.27% and others project USD 53.32 billion by 2035 Stem Cells Market Size To Hit USD 53.32 Bn By 2035—this dispersion highlights a single strategic truth: control of GMP-scaled, quality-assured production and cell-identity verification will determine which players capture commercial value over the next five to ten years.
39 days ago, we last updated this report. Notice something that’s not right? Let’s fix it together.
Topic Dominance Index of Stem Cell Biology
To gauge the influence of Stem Cell Biology within the technological landscape, the Dominance Index analyzes trends from published articles, newly established companies, and global search activity
Key Activities and Applications
- Allogeneic iPSC manufacturing and universal-donor lines — building immune-engineered, off-the-shelf starting materials to replace costly autologous workflows; this trend underpins the large strategic investments in iPSC platforms and HLA engineering.
- Directed differentiation for therapeutic endpoints — reproducible production of specialized cells (cardiomyocytes, neural subtypes, pancreatic beta cells) for transplantation and grafts; patents and clinical programs emphasize precise lineage control and lineage-specific protocols.
- Cell-based disease modeling and drug discovery — using iPSC-derived organoids and human cells for high-throughput screens to reduce animal-model risk and accelerate preclinical candidate selection.
- Scale-ready biomanufacturing and CDMO services — automated closed-system bioreactors, 3D suspension culture, and CDMO partnerships that translate lab-scale protocols into GMP batches for trials and commercialization.
- Quality assurance and genomic stability services — in-process genomic integrity testing and clone selection to mitigate tumorigenicity and regulatory risk, now a required capability to progress pluripotent programs toward market.
Emergent Trends and Core Insights
- Manufacturing-first competition is displacing single-asset lab programs: investors reward entities that demonstrate reproducible CQAs and lower cost of goods, producing a market architecture that favors platform enablers over isolated therapeutic bets STEMCELL Technologies.
- AI-driven process control is moving from analytics into decisioning — live-cell imaging, predictive differentiation models, and ML-directed process adjustments accelerate reliable differentiation and reduce batch failure rates.
- Cell-free modalities and secretome therapeutics (exosomes, EVs) present a regulatory and logistics arbitrage: lower complexity versus whole-cell products and faster clinical pathways for some indications.
- Patent focus shifting to process IP — as core growth-factor and pluripotency biology patents mature, the competitive edge moves toward high-efficiency differentiation protocols, purification methods, and automated QC workflows (as reflected in granted patents vs applications across key technique classes).
- Geographic split: North American commercialization, European mechanistic research, Asia manufacturing scale-up — market share and clinical activity concentrate where regulatory and capital environments align, but partnerships across regions remain critical for capacity and talent Stem Cells market synthesis.
Technologies and Methodologies
- CRISPR/Cas and advanced gene editing for immunoengineering — HLA editing and immune-silencing enable universal-donor constructs and reduce rejection risk Universal Cells.
- Deterministic cell programming (transcription-factor or inducible systems) — engineered, rapid differentiation platforms that shorten timelines from iPSC to functional cell types and improve batch consistency bit.bio.
- Automated closed-system bioreactors and 3D suspension culture — hardware and process innovations that scale production while reducing contamination and operator variability.
- Single-cell multi-omics and barcoding — high-resolution lineage mapping for clone selection and potency assays that directly address tumorigenicity and heterogeneity concerns Single cell analysis.
- AI-first live-cell imaging and predictive analytics — real-time phenotype classification to forecast differentiation outcomes and select clones with desired CQAs.
- Controlled-release growth-factor materials and synthetic, xeno-free media — consistent microenvironments and feeder-free systems that improve clinical-grade production reproducibility.
Stem Cell Biology Funding
A total of 123 Stem Cell Biology companies have received funding.
Overall, Stem Cell Biology companies have raised $10.4B.
Companies within the Stem Cell Biology domain have secured capital from 494 funding rounds.
The chart shows the funding trendline of Stem Cell Biology companies over the last 5 years
Stem Cell Biology Companies
- CellVoyant — CellVoyant pairs AI-first live-cell imaging with predictive models to forecast and optimise iPSC differentiation trajectories, aiming to reduce protocol iteration time and batch failure. Their platform targets early-stage developers and CDMOs seeking deterministic differentiation control and faster process transfer. This capability addresses a core bottleneck: inconsistent differentiation outcomes that hamper regulatory submissions.
- StemSight — StemSight develops iPSC-derived corneal therapies combined with functional biomaterials to simplify surgical delivery and improve graft integration, focusing on off-the-shelf products for corneal blindness indications. The company emphasizes scalable differentiation workflows and delivery simplicity to shorten clinical pathways in ophthalmology.
- Accellta — Accellta offers a 3D suspension production platform designed to cut manufacturing cost of pluripotent stem cells by orders of magnitude and accelerate throughput for cultured-tissue and CDMO applications. Their approach targets high-volume use cases such as cultured fat and regenerative tissue constructs, addressing the COGS challenge for therapeutic and alternative-protein markets.
- Stem Genomics — Stem Genomics provides in-process genomic stability testing and karyotyping solutions tailored to pluripotent stem cell manufacturing, shrinking regulatory risk tied to genomic anomalies and enabling routine clone screening at scale. Their assays directly respond to increasing regulatory expectations for genomic traceability in clinical-grade lines.
- Mesenbio — Mesenbio focuses on producing consistent extracellular vesicle therapeutics from immortalized MSC lines for musculoskeletal indications, positioning cell-free EV products as lower-complexity, lower-logistics alternatives to whole-cell therapies. Their strategy targets markets where exosome delivery can capture benefit without full cell-transplant regulatory burdens.
Get detailed analytics and profiles on 344 companies driving change in Stem Cell Biology, enabling you to make informed strategic decisions.
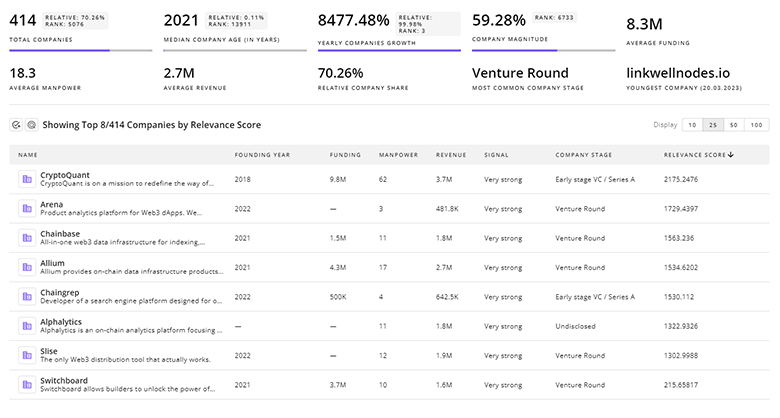
344 Stem Cell Biology Companies
Discover Stem Cell Biology Companies, their Funding, Manpower, Revenues, Stages, and much more
Stem Cell Biology Investors
TrendFeedr’s Investors tool provides an extensive overview of 723 Stem Cell Biology investors and their activities. By analyzing funding rounds and market trends, this tool equips you with the knowledge to make strategic investment decisions in the Stem Cell Biology sector.
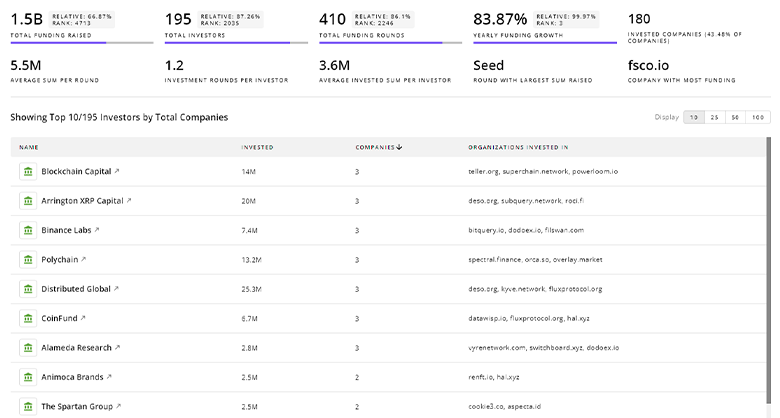
723 Stem Cell Biology Investors
Discover Stem Cell Biology Investors, Funding Rounds, Invested Amounts, and Funding Growth
Stem Cell Biology News
Explore the evolution and current state of Stem Cell Biology with TrendFeedr’s News feature. Access 1.5K Stem Cell Biology articles that provide comprehensive insights into market trends and technological advancements.
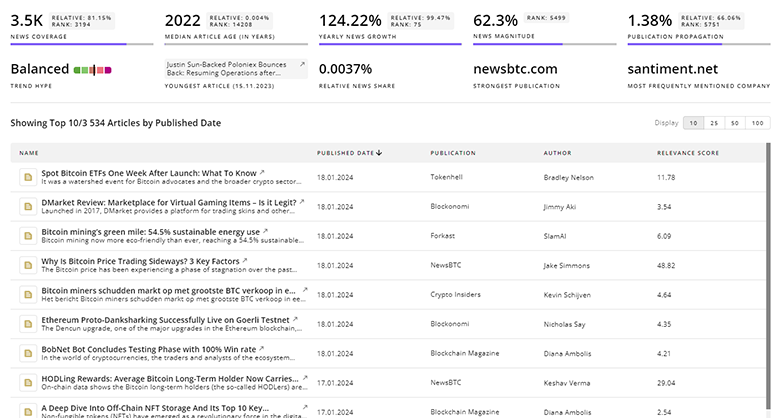
1.5K Stem Cell Biology News Articles
Discover Latest Stem Cell Biology Articles, News Magnitude, Publication Propagation, Yearly Growth, and Strongest Publications
Executive Summary
The strategic imperative for investors and developers in stem cell biology is clear: prioritize demonstrable, GMP-grade process control, scalable manufacturing, and rigorous QC capabilities before expanding therapeutic claims. Firms that provide deterministic differentiation, genomic surveillance, and automated production will become the backbone suppliers for clinical developers. Meanwhile, therapeutic teams should pair their biological claims with credible manufacturing pathways or partner with CDMOs and platform vendors early; doing so will shorten regulatory timelines and materially reduce commercial risk. Focused bets on cell-free modalities and on AI-driven process control offer asymmetric returns because they reduce regulatory friction and improve batch economics, respectively.
We're looking to collaborate with knowledgeable insiders to enhance our analysis of trends and tech. Join us!